IV. HEALTH HAZARDS AND EFFECTS
Nitric acid and related compounds, such as IRFNA, have been used in the chemical industry and are known toxic substances. The occupational health and medical literature on IRFNA and nitric acid exposure notes the general dangers and effects of nitric acid. However, IRFNA is a specific type of nitric acid, and the properties, hazards, and effects of any variety of nitric acid are similar:
When nitric acid is exposed to air or comes in contact with organic matter, it decomposes to yield nitric oxide and nitrogen dioxide, the latter being more hazardous than nitric acid. Exposure to high concentrations of nitric acid vapors and nitrogen oxides causes pneumonitis [inflammation of the lungs] and pulmonary edema [fluid build-up in the lungs, causing a lack of oxygen], which may be fatal; the onset of symptoms, such as dryness of the throat and nose, cough, chest pain, and dyspnea [labored breathing], may or may not be delayed. Prolonged exposure to low concentrations of the vapor may lead to chronic bronchitis and/or loss of appetite. The vapor and the mist may erode exposed teeth. However, in cases of dental erosion attributed to nitric acid, there was concomitant exposure to sulfuric acid, a potent cause of dental erosion.[63]
Most reported cases of severe illness from contact with nitrogen dioxide (one of the oxides in IRFNA fumes) derive from accidental exposures to explosion or combustion of nitroexplosives, nitric acid, the intermittent process of arc or gas welding (especially in a confined space), or entry into an unvented agricultural silo.[64] IRFNA in a military or wartime environment is not specifically addressed.
Given the ways in which Coalition servicemembers might have encountered IRFNA during and after the Gulf War, there are two general hazards associated with exposure: corrosion of skin and body tissue on contact with liquid IRFNA, and lung injury after inhalation of IRFNA and its oxides.[65] (See Figure 11.)
| TYPES
OF HAZARD/ EXPOSURE |
ACUTE
HAZARDS/ SYMPTOMS |
PREVENTION | FIRST
AID/ FIRE FIGHTING |
| Fire | Not combustible but enhances combustion of other substances. Gives off irritating or toxic fumes (or gasses) in a fire. | NO contact with flammable substances. NO contact with combustible or organic chemicals. | In case of fire in the surroundings: NO FOAM. |
| Explosion | Risk of fire and explosion on contact with many common organic compounds. | In case of fire: keep drums, etc., cool by spraying with water. | |
| Exposure | AVOID ALL CONTACT! | ||
| Inhalation | Burning sensation. Cough. Laboured breathing. Unconsciousness. Symptoms may be delayed. | Ventilation, local exhaust, or breathing protection. | Fresh air, rest. Half-upright position. Artificial respiration if indicated. Refer for medical attention. |
| Skin | Corrosive. Serious skin burns. Pain. Yellow discolouration. | Protective clothing. | Remove contaminated clothes. Rinse skin with plenty of water or shower. Refer for medical attention. |
| Eyes | Corrosive. Redness. Pain. Severe deep burns. | Face shield or eye protection in combination with breathing protection. | First rinse with plenty of water for several minutes (remove contact lenses if easily possible), then take a doctor. |
| Ingestion | Corrosive. Abdominal Pain. Burning sensation. Shock. | Do not eat, drink, or smoke during work. Wash hands before eating. | Do NOT induce vomiting. Give plenty of water to drink. Rest. Refer for medical attention. |
Figure 11. Exposure hazards of IRFNA and nitric acid[66]
On contact, IRFNA can cause corrosion to the skin and body tissue. The eyes are especially vulnerable to immediate damage following contact with the liquid or the concentrated vapor. Such contact may result in permanent eye damage with subsequent impairment of vision.[67]
Unlike the immediate effects of skin contact, symptoms from nitric acid vapor inhalation may be delayed, depending on the amount inhaled and the time over which the exposure occurs. These delayed symptoms include dryness of the throat and nose, cough, chest pain, dyspnea (labored breathing) and pneumonitis (inflammation of the lungs). Other symptoms of IRFNA inhalation can include headache, dizziness, lassitude (weariness or lethargy), nausea, vomiting, anxiety, choking, suffocation, and cyanosis (bluish or purplish discoloration of the skin or mucous membranes due to deficient oxygenation of the blood).[68]
In enclosed areas, IRFNA is particularly dangerous because the vapors are more contained and more easily inhaled. At low or moderate concentrations, IRFNA vapor inhalation may not cause any immediate symptoms, so that there may be few, if any, warning signs. As long as 24 hours after exposure to moderate or high levels, pulmonary edema can develop. Sometimes death may result due to complications from bronchopneumonia and other illnesses such as pulmonary fibrosis (scarring in the lungs).[69]
In open areas, the concentration of nitric acid vapors or mist normally declines rapidly, partly due to atmospheric diffusion. Nitric acid is highly hygroscopic, which means that it absorbs water vapor, causing the concentration level of the acid to drop rapidly.[70]
Because the Scud posed the most significant missile threat to Coalition forces, the CIA studied the IRFNA hazards from Scuds as a possible cause of Gulf War illnesses. The CIA’s study considered veterans’ Senate testimony, which reported their recollections of burning skin sensations after Scud impacts. The CIA assessed that these burning sensations were most likely caused by nitrogen dioxide fumes and droplets of IRFNA.[71]
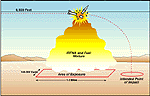
In sufficient quantity, these fumes and droplets can appear as a large reddish-brown cloud, persisting for about 30 minutes. For a cloud of this type to be visible, a Scud break-up or Patriot interception would have to occur below an altitude of three kilometers (approximately 9,500 feet). (See Figure 12, which is an artist’s representation of the CIA’s written study.) Below three kilometers, a Scud breakup or intercept would probably cause noticeable skin, eye, and respiratory effects on the ground. Otherwise, the IRFNA would be expected to dissipate into the atmosphere and not cause any hazard to people on the ground. The potential exposure area would probably be rather long and narrow—as large as two to three kilometers (approximately one to two miles) long by 100 to 200 meters (approximately 140 to 300 yards) wide.[72]
Figure 12. Area of exposure under a Scud break-up
The CIA also speculated about whether any IRFNA on Scud debris or in a hypothetical exposure area could reach lethal levels. It concluded that all the IRFNA on any Scud debris would probably evaporate in a matter of a few hours or less, making it a negligible threat to Coalition soldiers. On the other hand, exact IRFNA exposure levels on the ground would depend on:
How individuals might have been physiologically affected by such exposure would also be difficult to determine without professional medical diagnoses.
As described previously, permanent lung damage can result from IRFNA exposure. In addition, scars can also form from deep, slowly healing burns. The literature is less clear, however, on the possible chronic long-term effects of prolonged exposure to low concentrations of nitric acid vapor, and more particularly, nitrogen dioxide.[74] Though no chronic long-term health effects would be expected from a brief, one-time exposure to IRFNA, no specific medical literature discussion could be located.
A thorough search of the veterans-contact database in the Office of the Special Assistant, and a review of hundreds of interviews, revealed a few reports that may have been IRFNA exposures from mists contacting the skin and vapors inhaled near impacting Scud missiles. These veterans experienced symptoms which ranged from burning sensations in the eyes, nose, throat, and on the skin, to shortness of breath and temporary loss of voice. There were some reported exposures involving contact with debris from these missiles, but the reports of mist and vapor exposures were more frequent. Some cases required medical treatment, and some required hospitalization. However, there are no known cases of pulmonary edema from these potential exposures. There may have been less severe injuries from IRFNA exposure, but an analysis of the hospitalization statistics database showed only Center for Disease Control diagnosis codes; possible causes for hospitalization were not included in this database.
The veterans’ reports did confirm two incidents of skin burns from splattered, liquid IRFNA at the Kuwaiti Girls’ School Silkworm storage facility, where servicemembers had direct contact with the substance.[75] There was another reported incident involving direct IRFNA exposure--when a US Air Force EOD specialist disassembled a captured enemy missile.[76]
V. PROTECTION, MONITORING, AND DETECTION
The DoD developed its chemical warfare agent monitoring and detection measures around specific doctrine, equipment designs, and anticipated chemical warfare threats. During the Gulf War, some of the detection equipment produced false alarms for substances not really present—making the job of monitoring and detecting particularly daunting. The M8A1 Automatic Chemical Agent Alarm system, for example, registered false alarms for a multitude of reasons,[77] and there were many reports that M8A1 alarms sounded near Scud impacts.
Initially, IRFNA was not perceived as a threat. Military leaders did not expect large numbers of troops to be in contact with or near IRFNA (or weapon systems that used it). They anticipated a greater threat from the high-explosive warheads of incoming missiles (generally Scuds). The standard operating procedure for chemical and biological warfare agent defense was to take cover in designated shelters and to wear the protective mask and chemical protective suit as directed. EOD specialists sometimes wore special protective clothing and equipment (including self-contained breathing equipment) over their uniforms when working with hazardous materials.[79] (See Figure 13.)
Figure 13. A US Air Force EOD specialist, wearing breathing apparatus, special protective apron, and gloves, disassembles an enemy missile[78]
Commanders also issued general troop protection directives. On February 19, 1991, for example, the VII Corps sent a message emphasizing the importance of using the M256 Chemical Agent Detector Kit to confirm the presence of a chemical warfare agent. The VII Corps sent this message because of an incident five days earlier when US troops used a Chemical Agent Monitor or CAM (Figure 16) to test some impacted Scud debris. The CAM alarmed incorrectly for mustard agent, and its sensitive internal components were ruined.[80] The VII Corps’ chemical officer was aware of the toxicity of IRFNA and rocket propellants and concluded that propellant residue was a likely cause of the CAM failure.[81] The VII Corps therefore notified its subordinate units of the incident and provided instructions in the February 19th message, describing how to take measurements around a Scud impact site to ensure more reliable CAM readings.[82]
B. Chemical Monitoring and Detection Equipment
During the Gulf War US troops used chemical monitoring and detection devices designed to indicate the presence of enemy chemical warfare agents. Industrial monitors and detectors that could have indicated the presence of IRFNA were not deployed to the theater for the Gulf War. The Army’s only IRFNA monitoring and detection device was the multi-gas detector set, part of the Lance missile system, which was not used in the Gulf War. An Army EOD officer, who later served at the headquarters responsible for all Army EOD activities in the United States, confirmed that his teams did not carry the multi-gas detector kits, or any other device designed to indicate the presence of IRFNA.[83] It is highly unlikely, therefore, that other US EOD personnel had such detector kits available to them.
C. IRFNA Tests with Detection Equipment Used During the Gulf War
The sections which follow discuss how various chemical agent detectors react when exposed to IRFNA or related acids. Some of this information was gathered from Army field experiences, while information about other detector reactions was collected during laboratory tests at the Edgewood Research, Development, and Engineering Center (ERDEC), Aberdeen Proving Ground, Maryland, in January through March 1997. The ERDEC tests used laboratory red fuming nitric acid (without an inhibitor) because the testers could not acquire any rocket-grade red fuming nitric acid.
1. M8 and M9 Chemical Agent Detector Paper
M8 Chemical Agent Detector Paper (M8 paper) detects liquid chemical warfare agents. (See Figure 14.) Servicemembers carry a booklet of M8 paper and use it whenever they suspect liquid chemical agent contamination. To use the paper, the servicemember blots it on the surface of an object suspected of being contaminated. The paper turns dark green when it touches a V-type nerve agent, yellow when exposed to a G-type nerve agent, and red when it touches a blister agent.[84]
Figure 14. M8 Paper and M256A1 Chemical Detector Kit
M9 paper is the most widely used method of detecting liquid chemical warfare agents. It is more sensitive and reacts more rapidly than M8 paper.[85] The self-adhesive M9 paper attaches to most surfaces, while M8 paper cannot be attached to an item. Neither M8 nor M9 paper can detect chemical warfare agent vapor. M9 paper indicates the presence of a nerve agent or a blister agent by turning a red or reddish color,[86] but does not identify which agent gives the positive reading.
In the 1997 ERDEC testing, droplets of red fuming nitric acid on M8 paper caused dark brownish spots—not the characteristic green, yellow, or red spots normally caused by chemical warfare agents. Droplets of the red fuming nitric acid on M9 paper initially caused orange spots, which gradually turned golden—not the characteristic red spots normally caused by chemical warfare agents.[87]
2. M256A1 Chemical Agent Detector Kit
The M256A1 Chemical Agent Detector Kit is capable of detecting liquid and vapor concentrations of nerve, blood, and blister chemical warfare agents.[88] (See Figure 14.) It is issued to every ground force squad (the lowest organizational level, normally averaging 10 soldiers), so the squad can detect and classify chemical agents. The kit contains M8 paper for liquids and samplers for vapors.[89]
An analysis of the 1997 ERDEC laboratory test results shows that the M256A1 did not produce false indications for mustard or nerve agents when exposed to red fuming nitric acid. However, because of the acid content of RFNA, the M256A1 did sometimes falsely indicate the presence of a blood agent, hydrocyanic acid.[90]
3. M8A1 Automatic Chemical Agent Alarm System[91]
The M8A1 Automatic Chemical Agent Alarm System (M8A1) is an automatic chemical agent detection and warning system designed to detect nerve agent vapors and warn personnel with both audible and visual signals.[92] (See Figure 15.) The M8A1 system consists of the M43A1 Chemical Agent Detector Unit and the M42 Alarm Unit and is generally positioned up to 400 meters upwind of troop locations to provide early warning of chemical warfare attacks.[93]
Figure 15. Components of the M8A1 Automatic Chemical Alarm System
In the 1997 laboratory testing at ERDEC, testers exposed two M43 detectors to RFNA, and neither detector alarmed. The testers concluded that IRFNA would not set off the M8A1. However, IRFNA may disable the detector from alarming for any other agent. [94]
Given that the M8A1 alarmed in the presence of many substances other than nerve agents during Desert Storm, field observations about the M8A1 producing false positives might seem to contradict ERDEC’s conclusion. However, to-date there are no documented instances or warnings from the Gulf War indicating how the M8A1 performed when exposed to IRFNA.
The Chemical Agent Monitor (CAM) is a hand-held vapor monitor for mustard and nerve agents, which can report only conditions directly in front of the nozzle assembly. (See Figure 16.) It cannot give a realistic assessment of the vapor hazard over an area unless the operator moves the CAM around the area and takes several readings.[95]
Figure 16. Chemical Agent Monitor
As mentioned above, the VII Corps chemical office cautioned field units about using the CAM around missile debris in a February 19, 1991 message. The message stated that "…fuming nitric acid will drive the CAM to 8 bars on the mustard scale and will burnout the CAM if left for more than a few seconds at that level."[96] The message also recommends to its subordinate field units that the CAM not be held over missile debris for more than a few seconds. It also directs that any CAM detection be confirmed with the M256A1 kit.[97] Laboratory tests with red fuming nitric acid and the CAM at ERDEC in 1997 did not cause false positive readings for chemical warfare agents.[98] The ERDEC tests used red fuming nitric acid from a laboratory, which was not likely to contain the impurities of Iraqi military rocket-grade IRFNA. The ERDEC tests could not duplicate the VII Corps CAM readings or determine what fumes burned out the VII Corps CAM.[99]
| First Page | Prev Page
| Next Page |